deciphering the discipline
A regular column offering the student perspective of the next generation of ceramic and glass scientists, organized by the ACerS President’s Council of Student Advisors.

Graciela Martinez
Guest columnist
Facing the lithium shortage
Due to properties such as high thermal and electrical conductivity, low density, and high electropositivity, lithium is a key material used in numerous industries, including the ceramics and glass industry, air conditioning, greases, dental prostheses, various alloys, and medicines. Electronics are the cornerstone of the lithium market, with about 59% of global lithium production being used to fabricate lithium-ion batteries (LIBs) for electric vehicles, phones, tablets and other devices.1
The demand for this element is expected to increase dramatically in the coming years as governments and countries fast track the transition to green energy. But there are some concerns related to the supply of this element (see “Lithium: The 21st century ‘gold’ rush” in this issue). Considering these concerns, now is a good opportunity for science to make improvements in both the extraction and application of lithium.
From the extraction point of view, many scientists are investigating how to increase the purity of extracted lithium while minimizing the environmental impact of the extraction process. Traditional extraction techniques, such as pyrometallurgy or hydrometallurgy, are known for using excessive amounts of water, which become contaminated with base metals, such as cobalt.1
Lithium can also be obtained by recycling LIBs, which generally consists of a combination of pyrometallurgy, hydrometallurgy, and physical processes, among others. Obtaining lithium from LIBs has several drawbacks, however, such as the batteries not being immediately available for recycling, their transport and dismantling are expensive, and processing must be done carefully due to the high risk of explosion.1
Recently, other techniques like resynthesis or the use of microorganisms to extract base metals from spent LIBs have also been incorporated. These techniques, in addition to being environmentally friendly, are low cost. They still face some challenges to commercialization, including their long duration, low recovery, and the sensitivity of the microorganisms to temperature or pH.1
In terms of applications, scientists are investigating other materials that can replace lithium in batteries, such earth-abundant elements like sodium, potassium, magnesium, and aluminum. Studies have shown that batteries based on these elements can achieve similar performance to LIBs but at a lower cost.
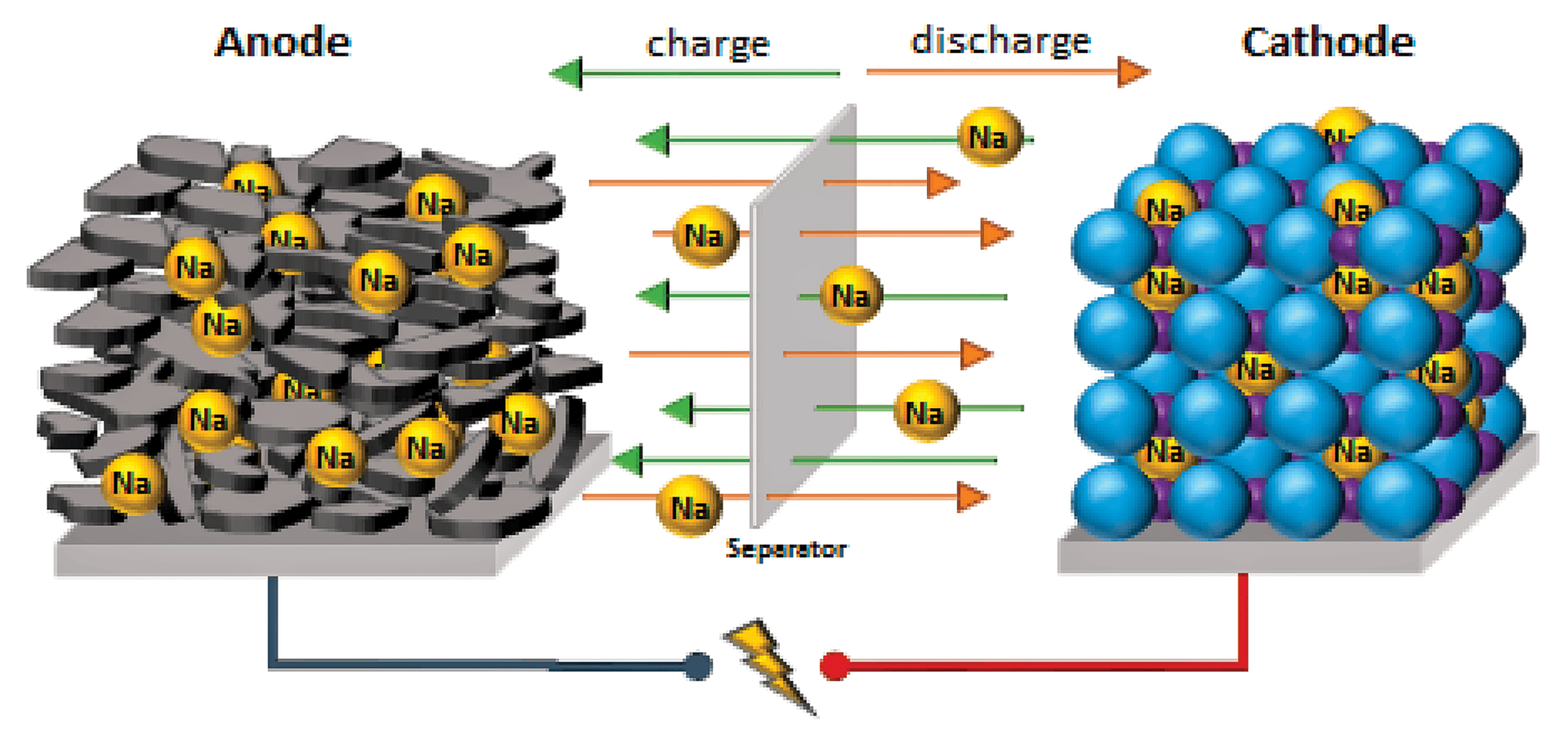
Illustration of a sodium-ion battery, one of the alternative battery compositions that may replace lithium.3
Credit: Shaylee Traugh
There are challenges to commercializing these alternative battery chemistries. For example, although very efficient cathodes have been found for sodium-ion batteries, researchers are still searching for appropriate anode materials that will not form dendrites. On the other hand, magnesium and aluminum-based batteries do not suffer from dendrite formation, but it is difficult to find cathode materials to match them. The small size of these ions makes their insertion or extraction difficult, resulting in a very strong coulombic interaction with the host cathode material, which leads to high charge/discharge rates.2
The options discussed above represent just a few of the many ways to tackle the lithium shortage. Researchers will continually find aspects that they can improve and problems that they can study and solve.
About the author:
Graciela Martinez is a master’s student at the Instituto Tecnológico de Saltillo in Mexico. Her graduate research involves developing advanced ceramics for solid oxide fuel cells and thermal barrier coatings in collaboration with the University of Castilla-La Mancha, Spain. In her free time, she enjoys playing video games and playing the drums.
AUGUST 2023 • VOL. 102, NO. 6

