deciphering the discipline
A regular column offering the student perspective of the next generation of ceramic and glass scientists, organized by the ACerS President’s Council of Student Advisors.

Carter Glynn
Guest columnist
From slimes to sun power: Looking at tellurium recovery from copper residue
With the rush toward renewable energy, the need for materials to support this transition has exploded. One of these materials is tellurium, an element rarer than gold or platinum in the Earth’s crust (0.001 ppm versus 0.004 ppm and 0.005 ppm, respectively).
More than half of the world’s tellurium is used in the form of cadmium telluride (CdTe), a direct bandgap material for thin-film solar technology. CdTe solar cells have continued to gain market share in the solar industry due to their greater energy efficiency and lower production costs compared to silicon solar cells.1
More than 95% of the world tellurium supply is a byproduct of copper production. Copper production involves casting mined copper concentrates and recycled copper scrap into anodes, which are then electrorefined to increase the purity of the copper (Figure 1).2 During the electrorefining process, impurities within the anode form fine black solids, or “slimes,” that sink to the bottom of an electrorefining cell. The slimes are collected and sent through several processing methods to recover numerous materials, including tellurium (>1 wt.%).
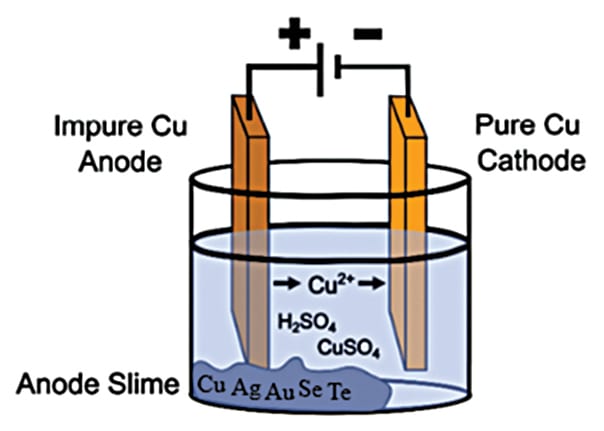
Figure 1. Simplified copper electrorefining cell displaying the formation of high-purity copper from cast copper anodes. An electrochemical reaction dissolves the anode to form copper ions and slimes. The slimes sink to the bottom of the cell while the copper ions deposit onto the cathode to form pure copper.
Credit: Ngan et al., Reviews in Environmental Science (CC BY 4.0)
Figure 1. Simplified copper electrorefining cell displaying the formation of high-purity copper from cast copper anodes. An electrochemical reaction dissolves the anode to form copper ions and slimes. The slimes sink to the bottom of the cell while the copper ions deposit onto the cathode to form pure copper.
Credit: Ngan et al., Reviews in Environmental Science (CC BY 4.0)

The first step in tellurium recovery is a process called decopperization. The most common decopperization method involves a pressurized sulfuric acid leach with oxygen to remove copper from the slimes by dissolution. During this process, tellurium oxidizes and forms tellurous acid (H2TeO3), which enters the leachate along with the copper. Copper and tellurium are then separated from the solution by adding solid copper to the leachate to form solid Cu2Te.
The amount of tellurium recovered through decopperization is inconsistent. Literature suggests that the recovery of tellurium can vary from 60–80% depending on many factors, including pretreatment of the slimes, starting composition, and processing conditions of the autoclave leach.3 Additionally, tellurium is known to reprecipitate during extended leaching times.4 Due to these factors, as well as the hurdles to establishing a consistent recovery process in copper refineries, currently only 29% of the tellurium present in copper anode slimes is recovered.5
To increase recovery rates, research groups have worked to characterize the slimes and optimize the autoclave leach process to understand how temperature, partial pressure of oxygen, sulfuric acid concentration, and solid-to-liquid ratios affect metal recovery. However, the impact of slime compositions and leaching time on tellurium extraction during pressure leaching has not been fully investigated.
My research at Missouri University of Science and Technology will focus on the impact of slime composition and leaching time on metal recovery rates. Regarding slime composition, my group plans to process slimes collected from multiple refineries under similar leaching conditions to observe how the recovery of tellurium can change based on the starting composition of the slimes.
Additionally, trials of extended leaching times are planned to investigate how tellurium may reprecipitate out of the leachate solution. Reprecipitation of tellurium after oxidation from tellurous acid to telluric acid (H2TeO4) is known to occur industrially, but it is not well reported in scientific journals. Better understanding of the precipitation process could help in identifying what tellurium compounds form along with what additions can be made to the slimes or leaching process to prevent this occurrence.
Ultimately, the results from these studies will improve tellurium recovery, thus helping fulfill the need for these valuable materials in the renewable energy transition.
Mx.
About the author:
Mx
Figure 1. The compositional complexity, structure, and site disorder of A6B2O17 disordered oxides make these materials attractive as potential high-entropy electroceramics. Adapted from Reference 4.
Credit: Spurling et al., J. Mater. Sci.
Pxx.
Credit: xx
About the author:
Carter Glynn is a master’s student in the group of professor Michael Moats at Missouri University of Science and Technology. His work focuses on the recovery of tellurium from copper anode slimes. In his spare time, he enjoys rock climbing and mountain biking.
