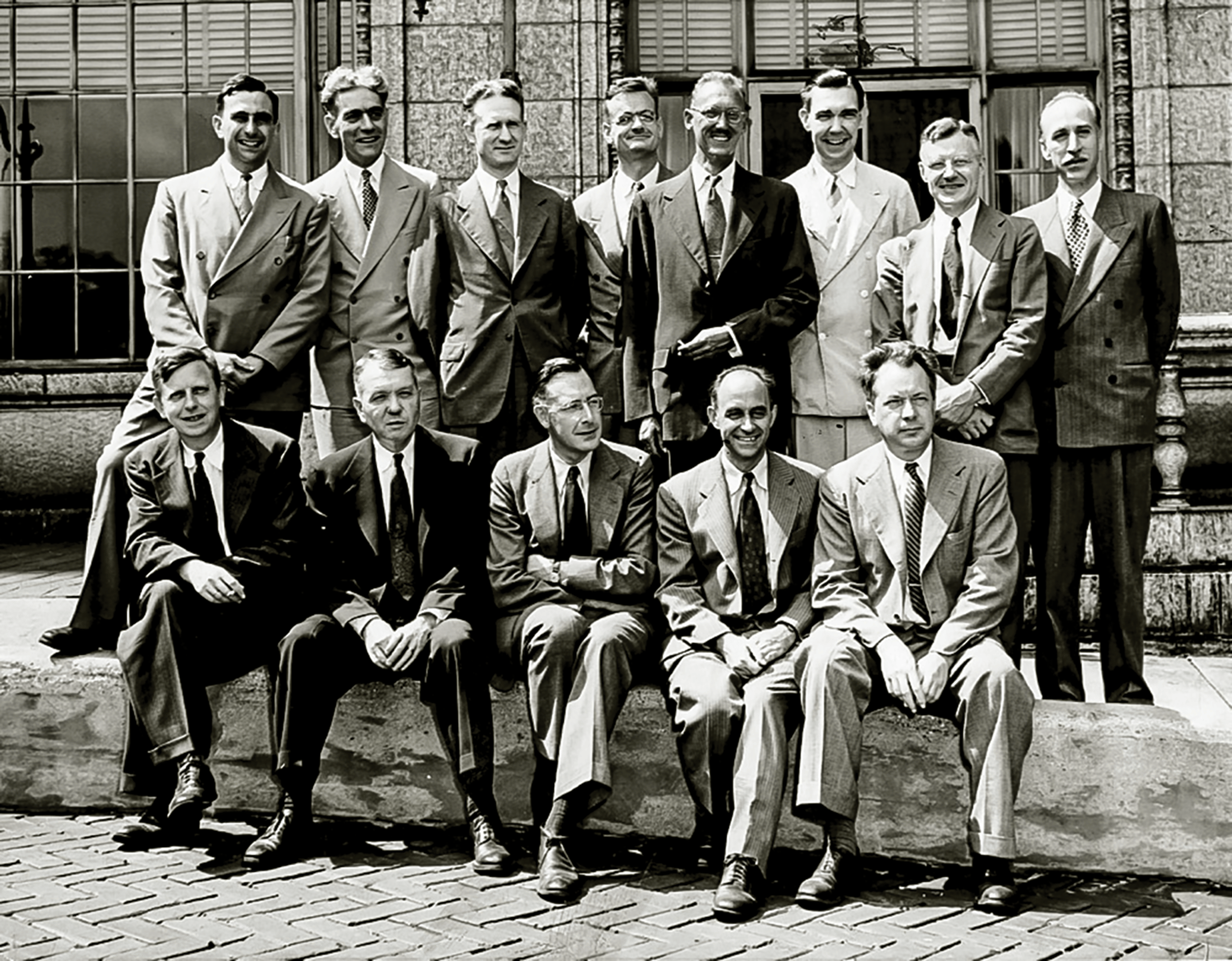
bulletin | feature

Numerous ceramists played hidden but key roles in the Manhattan Project. This article remembers the work of one such scientist, William Houlder Zachariasen.
By Mario Affatigato
Zachariasen and the Manhattan Project
Numerous ceramists played hidden but key roles in the Manhattan Project. This article remembers the work of one such scientist, William Houlder Zachariasen.
By Mario Affatigato
The popular movie “Oppenheimer” has provided a new generation of the U.S. public with a powerful recount of the events surrounding the Manhattan Project and the end of the Second World War.
In September 2023, Lisa McDonald—in a Ceramic Tech Today article—highlighted the role of Massachusetts Institute of Technology ceramists in the Manhattan Project,1 based on the work of Whittemore and McCreight. In this article, we seek to remember the work of William Houlder Zachariasen, a multifaceted physicist who made a seminal contribution to glass science and played an important role in the Manhattan Project.
Zachariasen was born in Langesund, Norway, in 1906. The town, near Oslo, gave Zachariasen (known as “Willie” to many) access to the fjord and the many islands there. This location is especially relevant because these islands had many mines rich in rare earths, sparking an early interest in mineralogy for Zachariasen. He studied at the University of Oslo under the guidance of the great geochemist Victor Moritz Goldschmidt,2,3 focusing on the X-ray characterization of minerals. Incredibly, Goldschmidt owned one of the islands and had a picnic there with Einstein in 1920!
After publishing his first paper in 1925, Zachariasen published 19 more papers before receiving his Ph.D. in 1928 at the age of 22. He followed this accelerated pace with a postdoctoral position in the lab of Nobel laureate Sir Lawrence Bragg, an expert on X-rays and crystal structure. During this one-year postdoctoral work, Zachariasen focused on silicates, perhaps impacting his later insights on glass structure.
After the postdoctoral fellowship, Zachariasen returned to the University of Oslo. Within a year (and upon Bragg’s recommendation), University of Chicago professor Arthur Compton offered Zachariasen a physics professorship at UChicago, which he accepted. He would stay at UChicago for 44 years.
Zachariasen was 24 years old when he left Norway in 1930. The early years in Chicago were tough. He had little money for research. Going to conferences, he would sleep on trains—coming and going on the same day—to save the hotel room cost. Using his prior work on silicates, he published his first (and only) paper on glass, “The atomic arrangement in glass” (1932). Yet this publication arguably became the most influential paper in the history of glass science. As Alfred R. Cooper wrote in his introductory paper to the 1980 Borate Glass Conference,4
“We dedicate this session on glass structure to Frederik William Holder Zachariasen because his single contribution to glass literature, ‘The atomic arrangement in glass,’ may be the most influential paper on glass structure in this century.”
Yet Zachariasen had no further interest in glass. Later in life, he had to be reminded that he published in this subject.
By 1943, Zachariasen had joined the Manhattan Project after becoming a U.S. citizen in 1941. Why was he needed? Fermi, working on the nuclear pile at UChicago, had demonstrated the chain reaction of uranium. Zachariasen was already involved in UChicago’s Metallurgical Laboratory. In the words of Robert A. Penneman,3
“Plans were being rushed for the pilot plant at Oak Ridge, Tennessee, and the production reactors at Hanford, Washington. This meant that the new element, plutonium, would be made in large quantity using neutrons from a nuclear reactor. Before this, plutonium could be made only in microgram quantities by tedious cyclotron irradiation.”
But plutonium metal had to be separated from uranium. Under Glenn Seaborg’s leadership, 60 chemists were put on this challenge. Zachariasen, with his expertise on rare earths, was to provide the X-ray analysis and “… deciphering single-handedly the composition of countless samples that were prepared by the chemists.”3
Seaborg wrote that on June 21, 1944, a 10-microgram sample that was thought to be neptunium dioxide (NpO2) was sent to Zachariasen. By 11 a.m. on June 22, his X-ray analysis had confirmed that it was NpO2. In his report, Zachariasen wrote:3
“The radius of Np+4 is thus 0.0158 Å larger than that of Pu+4, 0.016 Å smaller than that of U+4, and nearly identical with that of Ce+4. I believe that a new set of ‘rare earth’ elements has made its appearance. I believe that the persistent valence is four, so that thorium is to be regarded as the prototype just as lanthanum is the prototype of the regular rare earth elements.”
In a later interview, Zachariasen noted how hard the work was:
“We had a very exciting time struggling with all these patterns over the various plutonium compounds, identifying what the chemists had made and, hence, getting information about the chemistry of plutonium that was essential ... I remember working like hell on New Year’s Day and all holidays: often I worked late for many, many hours to get the work done. I had a wonderful time...”
Yet Zachariasen had no further interest in glass. Later in life, he had to be reminded that he published in this subject.
By 1943, Zachariasen had joined the Manhattan Project after becoming a U.S. citizen in 1941. Why was he needed? Fermi, working on the nuclear pile at UChicago, had demonstrated the chain reaction of uranium. Zachariasen was already involved in UChicago’s Metallurgical Laboratory. In the words of Robert A. Penneman,3
“Plans were being rushed for the pilot plant at Oak Ridge, Tennessee, and the production reactors at Hanford, Washington. This meant that the new element, plutonium, would be made in large quantity using neutrons from a nuclear reactor. Before this, plutonium could be made only in microgram quantities by tedious cyclotron irradiation.”
But plutonium metal had to be separated from uranium. Under Glenn Seaborg’s leadership, 60 chemists were put on this challenge. Zachariasen, with his expertise on rare earths, was to provide the X-ray analysis and “… deciphering single-handedly the composition of countless samples that were prepared by the chemists.”3
Seaborg wrote that on June 21, 1944, a 10-microgram sample that was thought to be neptunium dioxide (NpO2) was sent to Zachariasen. By 11 a.m. on June 22, his X-ray analysis had confirmed that it was NpO2. In his report, Zachariasen wrote:3
“The radius of Np+4 is thus 0.0158 Å larger than that of Pu+4, 0.016 Å smaller than that of U+4, and nearly identical with that of Ce+4. I believe that a new set of ‘rare earth’ elements has made its appearance. I believe that the persistent valence is four, so that thorium is to be regarded as the prototype just as lanthanum is the prototype of the regular rare earth elements.”
In a later interview, Zachariasen noted how hard the work was:
“We had a very exciting time struggling with all these patterns over the various plutonium compounds, identifying what the chemists had made and, hence, getting information about the chemistry of plutonium that was essential ... I remember working like hell on New Year’s Day and all holidays: often I worked late for many, many hours to get the work done. I had a wonderful time...”
Zachariasen was also a signatory (see middle column in Figure 1) to the Szilard Petition. The petition, drafted in July 1945, asked President Harry S. Truman to inform Japan of the terms of surrender before using atomic weapons. After much discussion (with Oppenheimer opposing it), the petition was brought to soon-to-be-named Secretary of State James F. Byrnes. He also was not sympathetic, and, therefore, President Truman never saw the petition.
Late in 1945, Zachariasen was appointed chair of the physics department at UChicago, and he also published a book titled “The Theory of X-Ray Diffraction in Crystals.” He was chair from 1945 to 1950, until an untimely heart attack, and then again from 1955–1959. Under his leadership, he brought Enrico Fermi, Edward Teller, Robert F. Christy, Walter H. Zinn, Maria Goeppert-Mayer, Gregor Wentzel, and other distinguished physicists to UChicago as professors of physics (Figure 2).
Figure 1. Scan of the Szilard Petition, which asked President Harry S. Truman to inform Japan of the terms of surrender before using atomic weapons.
Credit: National Archives and Records Administration

Zachariasen was also a signatory (see middle column in Figure 1) to the Szilard Petition. The petition, drafted in July 1945, asked President Harry S. Truman to inform Japan of the terms of surrender before using atomic weapons. After much discussion (with Oppenheimer opposing it), the petition was brought to soon-to-be-named Secretary of State James F. Byrnes. He also was not sympathetic, and, therefore, President Truman never saw the petition.
Late in 1945, Zachariasen was appointed chair of the physics department at UChicago, and he also published a book titled “The Theory of X-Ray Diffraction in Crystals.” He was chair from 1945 to 1950, until an untimely heart attack, and then again from 1955–1959. Under his leadership, he brought Enrico Fermi, Edward Teller, Robert F. Christy, Walter H. Zinn, Maria Goeppert-Mayer, Gregor Wentzel, and other distinguished physicists to UChicago as professors of physics (Figure 2).
Figure 1. Scan of the Szilard Petition, which asked President Harry S. Truman to inform Japan of the terms of surrender before using atomic weapons.
Credit: National Archives and Records Administration


Figure 1. Scan of the Szilard Petition, which asked President Harry S. Truman to inform Japan of the terms of surrender before using atomic weapons.
Credit: National Archives and Records Administration
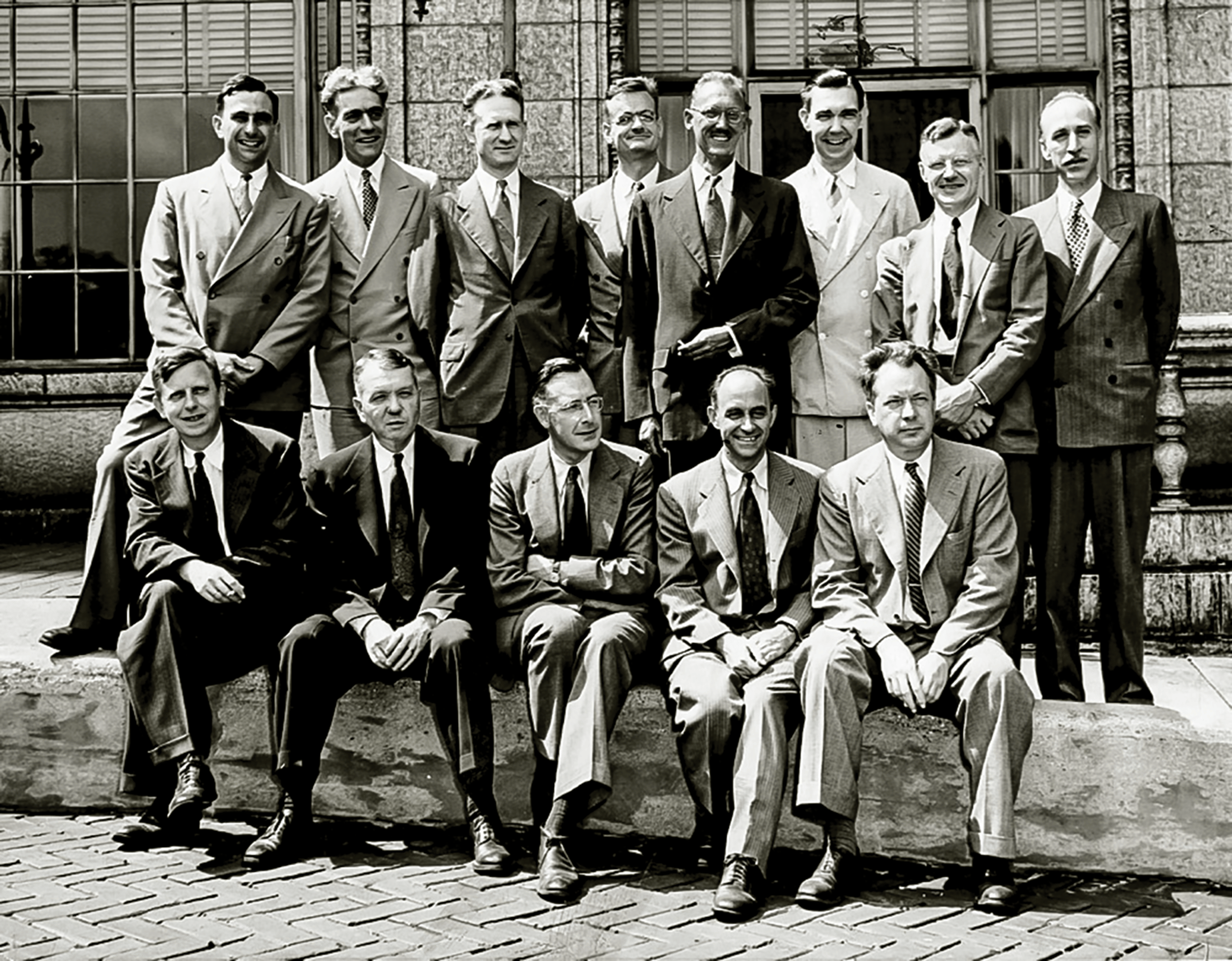
Figure 2. Seated (from left) Zachariasen, Urey, Smith, Fermi, Allison. Standing: Teller, Hogness, Zinn, Zener, Mayer, Schutz, Christ, and Eckart. Three were Nobel laureates: Zener created the Zener diode; Allison read the countdown to the Trinity test; and Zinn was the first director of Argonne National Laboratory.
Credit: University of Chicago Photographic Archive, [apf1-05555], Hanna Holborn Gray Special Collections Research Center, University of Chicago Library
During his time at UChicago and after, Zachariasen led a 37-year effort to elucidate the structure and chemistry of transuranic elements (metals and compounds). In 1948 alone, he published 26 papers. After retiring in 1970, Zachariasen consulted with friends at Los Alamos National Laboratory after moving to Santa Fe, N.M. He died on Dec. 24, 1979, aged 73.
Zachariasen’s contributions are memorialized in a variety of ways. The UChicago physics department holds one of its most significant lecture series in his name. But perhaps the most notable commentary on Zachariasen’s work was given by Linus Pauling (Nobel Laureate in Chemistry, 1954), who wrote:
“I have known Professor Zachariasen for nearly fifty years. His principal field of work has been the determination of the structure of crystals of inorganic substances by use of the X-ray diffraction technique. This is a field in which I have also done a large amount of work, and I believe that I am in a position to form a sound opinion about his ability and his contributions. It is my opinion that he has been and is the world leader in this field. I feel that he is to be classed among the outstanding scientists of the twentieth century, and at the top of the field in inorganic crystal structures.”
About the author:
Mario Affatigato is the Fran Allison and Francis Halpin Professor of Physics at Coe College in Cedar Rapids, Iowa. Contact Affatigato.
